Structural insight of a photosystem I-CpcL-phycobilisome supercomplex from a cyanobacterium Anabaena sp. PCC 7120
Zhiyuan Mao, Zhenhua Li, Xingyue Li, and Guangye Han
PNAS; March 25, 2026; 123 (13) e2530459123; https://doi.org/10.1073/pnas.2530459123
Significance
The light reactions of oxygenic photosynthesis are powered by Photosystem I (PSI) and Photosystem II (PSII), which consist of a core and the peripheral light-harvesting antenna complexes. In cyanobacteria, phycobilisomes (PBSs) serve as the main light-harvesting antenna. A rod-type PBS specifically binds to PSI through the CpcL linker protein, forming a PSI-CpcL-PBS supercomplex. Here, we solved cryo-EM structures of PSI-CpcL-PBS and CpcL-PBS supercomplexes from a cyanobacterium Anabaena sp. PCC 7120. These structures reveal the assembly of 18 pairs of phycocyanin αβ monomers and linker proteins (CpcL, CpcC1, CpcC2, and PecC) as well as the attachment of CpcL to PSI. These results provide insights into the organization of the supercomplex and the light capture and energy transfer mechanisms within this supercomplex.
Abstract
Phycobilisomes (PBSs) are supramolecular pigment–protein complexes composed of phycobiliproteins and linker proteins, serving as the major light-harvesting complexes that capture and transfer light energy to photosystem II (PSII) and photosystem I (PSI) in cyanobacteria and eukaryotic red algae. In cyanobacteria, a rod-type PBS that does not have a core is specifically connected to PSI by a linker protein CpcL to form a PSI-CpcL-PBS supercomplex. However, the mechanism of CpcL-PBS association to PSI remains unclear. Here, we report the cryoelectron microscopic structures of PSI-CpcL-PBS at 2.98 Å and CpcL-PBS at 2.93 Å resolution from a cyanobacterium Anabaena sp. PCC 7120, respectively. CpcL-PBS is located on the stromal side of a PSI tetramer and exhibits a structure of three-layered PBS consisting of four linkers (CpcL, CpcC1, CpcC2, PecC) and 18 pairs of phycocyanin αβ monomers. The C-terminal transmembrane helix of CpcL inserts to the membrane and interacts with PsaA, PsaB, and PsaM of PSI at an interface I between two PSI monomers, enabling the formation of the PSI-CpcL-PBS supercomplex. The exact structure of protein subunits and arrangement of bilin and chlorophyll pigments are revealed, which provide a structural basis for the assembly of PSI-CpcL-PBS and possible excitation energy transfer pathways from antennas to PSI within this supercomplex, shedding light on the organization and attachment of CpcL-PBS in cyanobacterial thylakoids.
See: https://www.pnas.org/doi/10.1073/pnas.2530459123
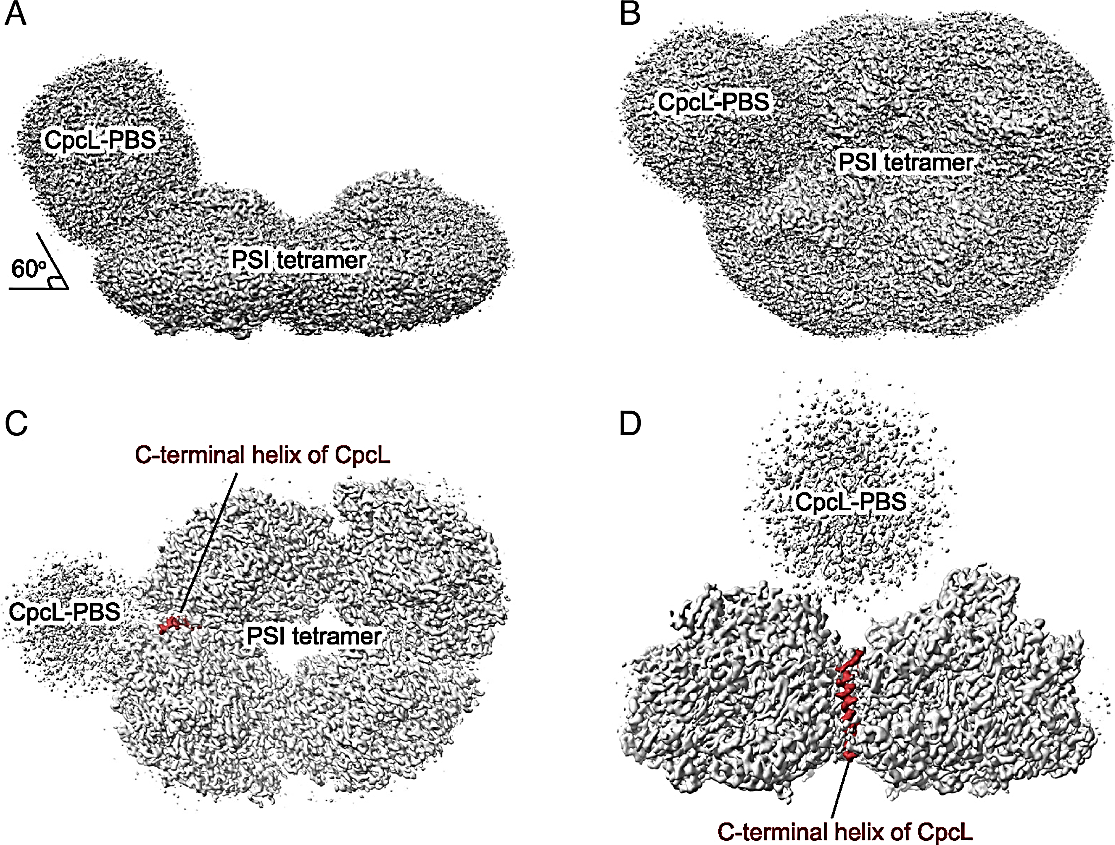
Figure 1:
Overall cryo-EM density map of the PSI-CpcL-PBS supercomplex from Anabaena sp. PCC 7120. (A) Side view of the cryo-EM density map of PSI-CpcL-PBS supercomplex, viewed along the membrane plane (threshold level = 0.1, Chimera). (B) Top view of the cryo-EM density map of PSI-CpcL-PBS supercomplex from the stromal side (threshold level = 0.1, Chimera). (C) Top view of the cryo-EM density map of the PSI-CpcL-PBS supercomplex from the stromal side at a higher threshold level (threshold level = 0.2, Chimera). (D) Side view of the cryo-EM density map of the PSI-CpcL-PBS supercomplex, viewed along the membrane plane with an angle different from panel A at a higher threshold level (threshold level = 0.2, Chimera). The C-terminal helix of CpcL is highlighted in brown.
Views: 65


