Pathogen-inspired engineering of plant protease enhances late blight resistance
Jie Huang, Alice Penrose, Laura Ossorio Carballo, and Renier A. L. van der Hoorn
PNAS; January 9 2026; 123 (2) e2524700123; https://doi.org/10.1073/pnas.2524700123

Significance
Genetic engineering to produce crops that are pathogen resistant is an important strategy for world food security. Here, we engineered tomato-secreted immune protease C14 to become less sensitive to inhibition by cystatin-like inhibitor extracellular protease inhibitors of cysteine proteases (EpiCs) secreted by the oomycete late blight pathogen Phytophthora infestans, and demonstrated that this engineered C14 (eC14) provides increased immunity. Importantly, this engineering was inspired by our identification of two proteases (Pain1 and Pain2) secreted by P. infestans that contribute to virulence and have reduced sensitivity to EpiCs inhibition. Thus, a pathogen avoiding self-inhibition can inspire crop engineering.
Abstract
The apoplast is an important battlefield in plant–pathogen interactions. The late blight oomycete pathogen Phytophthora infestans, for instance, secretes cystatin-like protease inhibitors EpiC1 and EpiC2B to suppress C14, a papain-like immune protease secreted by tomato. Here, we found that P. infestans also secretes two distinct papain-like proteases termed Pain1 and Pain2, which are transcriptionally induced during infection. Both Pains promote P. infestans infection, but not when their catalytic residues are mutated. Strikingly, EpiC1 and EpiC2B preferentially inhibit tomato C14 rather than self-produced Pains, suggesting that they coevolved with Pains to avoid self-inhibition. To mimic the avoidance of inhibition by EpiCs, we engineered C14 (eC14) with seven Pain1 residues that potentially disturb the EpiCs–C14 interface. This eC14 is less sensitive to inhibition by EpiCs and enhances resistance to P. infestans infection. This strategy demonstrates that a pathogen-inspired protein engineering approach can increase crop resistance to plant pathogens.
See https://www.pnas.org/doi/10.1073/pnas.2524700123
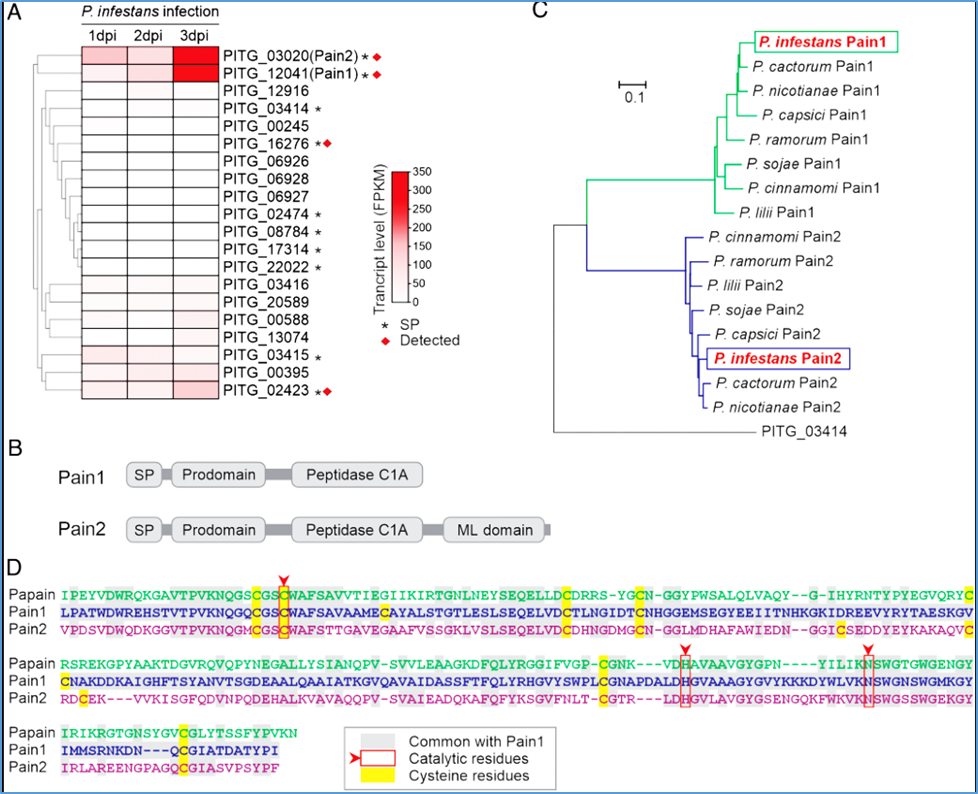
Figure 1:
Pain1 and Pain2 are induced and secreted during infection. (A) Gene expression profiles of 20 putative PLCPs of P. infestans. Ten PLCPs contain predicted signal peptides (asterisks), and four (red squares) have been detected in the P. infestans secretome (24). Gene expression data of 20 PLCPs are provided in SI Appendix, Table S5. (B) Domain architecture of Pain1 and Pain2. Both proteins contain a signal peptide, a prodomain, and a C1A protease domain. In addition, Pain2 possesses a C-terminal ML domain. (C) Phylogenetic analysis of Pains orthologs across Phytophthora species based on full-length protein sequences. Homologous sequences were identified in P. cactorum, P. nicotianae, P. capsici, P. lilii, P. ramorum, P. sojae, and P. cinnamomi. The secreted PLCP PITG_03414 from P. infestans was included as an outgroup. Sequences are provided in Dataset S2. (D) Sequence alignment of the protease domains of Pain1 with Papain (Top) and Pain2 (Bottom). Conserved catalytic residues are highlighted with red arrows and boxes.
Views: 61


