Rubisco kinetic acclimation at the holoenzyme level
Bryce Askey, Maddie Ceminsky, Elena Scott, Yongsheng Wang, Zhen Guo Oh, Stavros Azinas, Arthur Laganowsky, and Laura Helen Gunn
PNAS; April 15 2026; 123 (16) e2519914123; https://doi.org/10.1073/pnas.2519914123
Significance
Kinetic acclimation enables proteins to adjust their activity in response to environmental perturbations. For the CO2-fixing enzyme Rubisco, kinetic acclimation may be conferred by its small subunits. Plants express multiple small subunits and vary their expression with temperature. Here, we demonstrate that different small subunits can bind to the same Rubisco to form a heterogeneous holoenzyme. These small subunits had distinct kinetic effects which aligned with changes in holoenzyme structure and stability. Our findings indicate that small subunits enable Rubisco kinetic acclimation via manipulation of flexibility. By assembling a more rigid active site in higher temperatures and a more flexible one in lower temperatures, plants maximize the efficiency of their Rubisco, and thus photosynthesis, over a wide range of temperatures.
Abstract
In plants, the CO2-fixing enzyme Rubisco is hexadecameric, with each mature holoenzyme containing eight small subunits (SSus). Many plants express multiple SSus and vary their expression in response to environmental cues. Previous work indicates that this may allow fine-tuning of Rubisco’s performance in a variable environment (i.e., kinetic acclimation). Despite SSu pools being heterogeneous and dynamic, nearly no evidence exists for holoenzyme-level heterogeneity. Here, we characterized the structural and kinetic plasticity of Rubisco. We first established that SSu-heterogeneous Rubisco exists in Arabidopsis thaliana and quantified the prevalence of heterogeneity. We found SSu-heterogeneous Rubisco to make up over half of the Rubisco pool when heterologously expressed. This Rubisco contained at least four unique SSu ratios, indicating a variety of holoenzyme arrangements are possible. We then tested the kinetic effect of different SSus and found heterogeneity to have an antagonistic effect on substrate and inhibitor affinity. Kinetic differences between the SSus correlated with changes in local flexibility, and cryo-EM analysis illustrated a structural mechanism through which SSus may influence catalysis. Our kinetic and structural findings align with the hypothesized role of SSus in kinetic acclimation, as we observed the warm temperature-expressed SSu of A. thaliana to confer a stabilizing effect to the active site relative to the cool temperature-expressed SSu. This increase in stability manifested as a reduction in flexibility and increase in substrate affinity, indicating that fine-tuning of local stability may underlie Rubisco kinetic acclimation.
See: https://www.pnas.org/doi/10.1073/pnas.2519914123
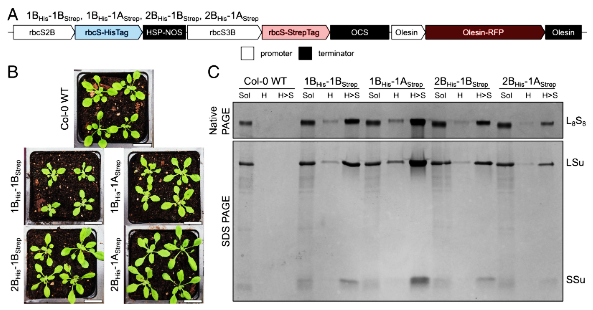
Figure 1:
SSu-heterogeneous Rubisco can be purified from A. thaliana plants. (A) Dual-SSu construct design used to generate transformed plant lines. (B) 3-wk-old T2 plants. (Scale bar, 2 cm.) (C) Native and SDS PAGE of total soluble and affinity purified protein from T3 plants. Rubisco bands in H > S lanes appear more intense than those in H lanes because H > S fractions were concentrated prior to loading. Sol: total soluble; H: HisTrap purified; H > S: HisTrap and StrepTrap purified; RFP: red fluorescent protein.
Views: 5


